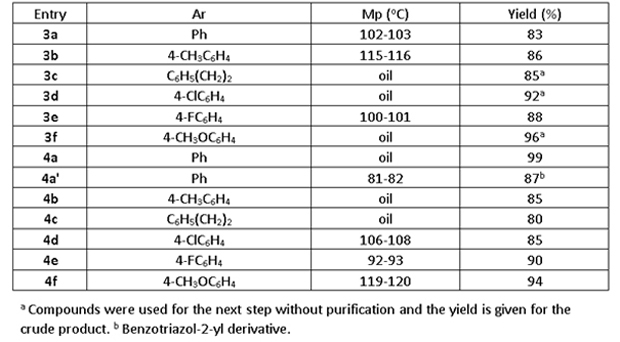
2.2.3 Title
The title (16 pt, bold, centered; followed by one empty line) should clearly describe the purpose of the paper and should not include abbreviations or acronyms. Only the first letter of the title and of proper names in the title (e.g., Lewis acid) should be capitalized.
2.2.4 Authors' name(s)
Typically, the following should be provided for each author:
Given name, middle initial (if any) and surname (12 pt, bold, centered; followed by one empty line).
Please note that the surname will be assumed to be the last word of the complete name, and will be indexed as such. The name of the corresponding author (the author submitting the manuscript, and responsible for dealings with the Editors) should be marked with an asterisk (*). If authors are from different institutions their surnames should be labeled after the name with a lower-case superscript letter referring to the authors' addresses (see
2.2.5). The last Author's name of the Author line (if more than one author) should be preceded by "and".
Example:
Albert J. Jones,a Dalpat Patel,b and Arthur Other*b
2.2.5 Authors' address(es), corresponding author's e-mail address
The complete mailing address(es) of author(s) should include the name of the institutional affiliation, street or P.O. box number, city, postal code, and country (12 pt, italic, centered).
The address line should be followed by the e-mail address of the corresponding author (12 pt, italic, centered; with hyperlink; followed by one empty line).
If two or more address lines are needed, they should be distinguished by superscript
letters preceding each address; the relevant letters should follow the authors' names in the author line (see 2.2.4 above).
2.2.6 If a
Dedication is desired, it should appear after a one line space below the e-mail address (12 pt, bold, centered).
2.2.7 Abstracts
Abstracts must be comprised of a written abstract and a graphical abstract.
2.2.7.1 Written Abstract
The written abstract should state the purpose of the research, the principal results and major conclusion(s). A well-written abstract will help to attract the attention of readers and, thus, increase the likelihood that the published article will be cited by other researchers. Abstracts should be comprehensible without inclusion of compound numbers and experimental results which are more suitable for the Results and Discussion. A typical abstract should contain no more than 100 words to fit in into the Abstract block with the Graphical Abstract.
Abstract: Heading: 12 pt, bold, left-aligned. Abstract text (on next line): 12 pt, normal, justified; 6-pt space above, in a single paragraph, followed by one empty line.
2.2.7.2 Graphical Abstract
A graphical abstract should immediately follow the written abstract to provide readers a diagrammatic impression of the chemistry described without being detailed. It is not intended to summarize the results. It should be created in ChemDraw (or ISIS Draw), should not contain structure numbers, and should be centered as a .cdx file and not as a picture image (See Section 2.4.1 under “Graphics”).
Authors: Please note that a separate graphical abstract is no longer required.
2.2.8 Keywords: The Abstract should be followed by four to six keywords.
Keywords subheading: 12 pt, bold, colon, followed on the same line by keywords (first one capitalized, and all separated by commas): 12 pt, Calibri (Body), left justified. There should be two-lines spacing before the next heading.
Content intended for the first page of the paper (including Keywords) MUST NOT be displaced onto page 2 due to the Abstract and Graphical Abstract being too lengthy or large, respectively.
2.2.9 Headings font size and style, alignment:
Major section headings (Introduction, Results and Discussion, Experimental Section, References, etc.): 14 pt, bold, left aligned, blue highlight background. There should be two spaces between sections and one space between each section heading and the text. The first paragraph of the text after major headings should not be indented.
Section subheadings, level 2: 12 pt, bold, left aligned. Text begins on the following line and should not be indented.
Section subheadings, level 3: 12 pt, bold, run on with first text paragraph, and so L/R justified.
Paragraphs should be indented (recommended 0.25 in or 0.7 cm) except the first paragraph after a section heading. In the Experimental Section paragraphs should not be indented.
2.2.10 Reviews and Accounts - Table of Contents (ToC)
A ToC should appear above the Introduction for Reviews and Accounts only. The headings should follow the hierarchy and style as indicated below. The Introduction should be numbered 1. The References list heading should not be numbered. In the ToC, all items should be 12pt, with no emphasis (bold or italic).
1. Primary heading (in text, 14pt bold, flushed left, blue highlight background)
1.1 Secondary heading single (1 cm) indent. (in text, 12pt bold, flushed left)
1.1.1 Tertiary heading, double indent (2 cm) (in text, 12-pt bold, run on with paragraph ("normal" style, no indent)
1.1.1.1 Quaternary heading (not recommended normally) (in text, italic, run on with paragraph)
2. Primary Heading
An introduction, which clearly states the thesis, objectives and relevance of the research, and any prior research, must begin at the top of the second page of the manuscript with the heading
Introduction on the first line, unless the paper is a review or an account, when it follows the
ToC.
2.2.12 Results and Discussion
All significant results that need to be discussed and rationalized should be contained in this section. Experimental details including Figures, Tables and Schemes should be kept to a necessary minimum, and reiteration of information from such should be avoided. Additional Tables, Figures and Schemes should be contained in a Supplementary Material file.
2.2.13 Conclusions
The Conclusion should summarize the main findings, including the significance of the results, and SHOULD NOT include compound numbers or other experimental information already presented in the Results and Discussion.
2.2.14 Experimental Section
General. This section should contain a description of instruments (make and model), and any special procedures used. The sources of all commercially available chemicals, the origin of all combustion analytical data, and the location of any work done outside the authors' institution, should be given.
Synthetic procedures should be accompanied by a listing of the most important product characterization data. Each paragraph describing a synthetic experiment should give the systematic (IUPAC) name of the product together with the number assigned to the compound in the Results and Discussion section. Use of standard abbreviations or unambiguous molecular formulae for reagents and solvents, and of structure numbers rather than chemical names to identify starting materials and intermediates, are encouraged. Reactant and reagent quantities should be given in both weight and molar units. Yields should represent weighed amounts of isolated and purified products and should be reported as percentages. For recrystallization, the solvent used should be identified and, in the case of chromatographic purification, both the eluting solvent and the type of support used should be given. In the preparation of a compound series, a general experimental procedure should be given
All spectra relevant to compound identity or purity must be submitted as supporting information in a Supplementary Material file.
Abbreviations, acronyms, symbols, and physical constants. Common abbreviations, acronyms, and symbols may be used, but less common ones should be defined the first time they are used (see the ACS Style Guide). Usage within a paper should be consistent, e.g., either Ph or C
6H
5 for phenyl, not both. Note: Ph should not be used for C
6H
4. The symbols used for physical constants should be in italics (e.g.,
d, J, k, c, m/z), but the units for physical constants should
not be italicized. Equal signs should be avoided in citing data, e.g.,
J 5.1 Hz not
J = 5.1 Hz.
QSAR studies. Arkivoc considers only those QSAR papers that meet the following criteria: (i) the work must involve statistical treatment of results obtained from the authors’ laboratory (they must not be culled from the published literature) and (ii) the paper should include a practical (experimental) element, as appropriate to an organic chemistry journal. See section 1 for the Arkivoc policy on medicinal chemistry papers.
Microwave reactors. Experiments conducted in microwave reactors must indicate whether sealed or open-reaction vessels were used and must document the manufacturer and model of the reactor, the method of monitoring the reaction mixture temperature (external surface sensor or internal probe), and the temperature reached or maintained in each experiment.
Please note that Arkivoc does not publish reports of studies conducted with domestic (kitchen) microwave ovens.
Photochemical work. In the case of light-promoted reactions, the details of the light and the experimental setup should be given. The nature and amounts of photocatalysts should also be stated. For work using UV irradiation, the material of the reaction vessel as well as the use of filters should be reported.
Compound characterization data. Arkivoc maintains the highest standards for compound characterization to ensure that compounds being added to the chemical literature have been correctly identified, and could be synthesized in expressed yields and purities using the reported preparation, isolation, and purification methods. For all
new compounds, evidence adequate to establish both
identity and
degree of purity (homogeneity) must be provided. Purity documentation must also be provided for
known compounds whose preparation by a new or improved method is reported.
If, in the opinion of the Scientific Editor, new compounds have not been satisfactorily characterized, the paper will be rejected.
For known compounds synthesized by published methods, the method of preparation, the literature data used to confirm the identity of the material, and the technique(s) used to determine purity should be reported. For known compounds prepared by new or modified synthetic reactions, the types of physical and spectroscopic data found to match cited literature data should be given, and purity documentation (see below) should be provided. Listings of IR and NMR absorptions and MS peaks for known compounds should be included only if they do not duplicate published data.
Evidence for the identity of new compounds must include
both 1H and
13C NMR spectral data
and either HRMS or elemental analysis data. A criterion of purity (see below) must also be provided with HRMS data.
The interpretation of NMR spectra, together with the applied method (Including 2D NMR spectra), is encouraged. Authors using automated data analysis are reminded to check numerical data (including proton counts, multiplicities and coupling constants) before including them in the manuscript. Coupling constants should match numerically for mutually coupled protons.
For products isolated as inseparable isomeric mixtures, the NMR chemical shift data for each isomer should be reported in two or more separate lists, one for each isomer, instead of as a single list. For proton NMR data, the integrals in each isomer's list should be reported in whole numbers of protons.
The resolution of the spectra should be high enough so that fine-structure multiplets can be viewed by increasing the image magnification (zoom). In cases where structure assignments of complex molecules depend heavily on NMR data interpretation, including isolated and synthesized natural products, copies of the 2D spectra should also be provided.
Proton and carbon NMR resonance signals should be listed for each new compound; the solvent and instrument frequency should be identified. Carbon NMR peak shifts should be rounded off to the nearest 0.1 ppm., proton spectra to the nearest 0.01 ppm. Hydrogen multiplicity (C, CH, CH
2, CH
3) information (APT) obtained from carbon DEPT or 2D spectra may be included with the carbon peak shifts, if available, but is not essential information. The
J values for all signals described as ‘d’, ‘t’, etc., must be given. For an example of the format for reporting NMR data, please see the model Experimental Section at the end of the Instructions to Authors.
For compounds synthesized by several methods, the purity documentation (typically a proton NMR spectrum in the supporting information) should clearly identify which method provided the sample, the purity of which is documented. The normal full range of chemical shifts should be displayed (usually 0-10 ppm for proton; 0-200 ppm for carbon).
For new compounds, copies of both proton and carbon NMR spectra are required and must be included in an accompanying Supplementary Material file.
For new compounds, either HRMS (resolution 0.001
m/z unit or better) or combustion elemental analysis data should be reported to support the molecular formula assignment. The reported data should include the molecular formulae on which the theoretical (Calcd) values are based.
HRMS molecular formulae and Calcd values should include any added atoms (usually H or Na). The ionization method should be reported, and a criterion of purity provided. A Found value within 0.003
m/z unit of the Calcd value of a parent-derived ion (for molecular weights less than 500), together with other available data (including knowledge of the elements present in reactants and reagents) is usually adequate for the unique determination of a molecular formula.
Elemental analysis or HMRS data are not required for enantio-enriched compounds characterized as racemates or for the second enantiomer when the synthesis and characterization of both enantiomers is reported. In these cases, however, the chemical and enantiomeric purities of each enantiomer must be documented, e.g., through chiral analytical chromatography. Where enantiomeric compositions of enriched mixtures are analyzed, the respective data (chromatograms, spectra, etc.) must be shown for the racemate.
Elemental analysis. Values found for carbon, hydrogen, and nitrogen (if present) should be within 0.4% of the Calcd values of the proposed formula. The inclusion of fractional molecules of solvent or water in the molecular formula to improve the fit of the data usually reflects incomplete purification of the sample. If the reported formula includes such a molecule, independent evidence for its presence (such as NMR peaks with appropriate chemical shifts) should be presented. The format for reporting CHN analyses can be found in the model Experimental Section at the end of the Instructions to Authors. The name and address of the analytical laboratory should be included in the General paragraph at the beginning of the Experimental Section.
Specific rotation. Specific optical rotations should be reported for isolated natural products, enantiopure compounds, and enantioenriched isomer mixtures when sufficient sample is available. Specific rotations based on the equation [α] = (100.α)/(
l.c) should be reported as unitless numbers as in the following example: [α]
D20 -25 (
c 1.9, CHCl
3), where the concentration
c is in g/l00 mL and the path length
l is in decimeters. The units of the specific rotation, (deg.mL)/(g.dm), are implicit and should not be included with the reported value.
Melting point. A melting point
range should be reported for every crystalline solid product, together with the solvent from which it was recrystallised, and a description of the material, e.g., "yellow needles, mp 123-124 °C (from EtOH)". For non-crystalline materials, there should also be a description, e.g., "brown oil; colorless gum". And, similarly, for liquids, e.g., "colorless oil, bp 123 °C (120 mmHg)".
IR and MS. Only those IR absorptions diagnostic for major functional groups, and only those MS peaks used for structure assignment, should be included in the experimental section. Complete peak listings or copies of the spectra may be included in the Supporting Information (Supplementary Material). IR bands, when reported, should be indicated using an approximate scale of relative intensities (vs, s, m, w = very strong, strong, medium, weak). Significant MS peaks should be reported with their % intensity relative to the base peak of the spectrum.
Purity. If a Scientific Editor feels it necessary to ask for evidence for compound purity, that evidence should include
one or more of the following:
1. A standard 1D proton NMR spectrum or proton-decoupled
13C NMR spectrum showing, at most, trace peaks not attributable to the assigned structure.
2. Combustion elemental analytical values for carbon and hydrogen (and nitrogen, if present) which agree with calculated values within 0.4%.
3. Quantitative gas chromatographic analytical data for distilled or vacuum-transferred samples, or quantitative HPLC analytical data for materials isolated by column chromatography or separation from a solid support.
4. For known solid compounds, a narrow melting point range that is in close agreement with a cited literature value.
Spectra. Reproductions of spectra should be published in the Results and Discussion section
only when concise numerical summaries are inadequate for the discussion. Papers dealing primarily with interpretation of spectra, and those in which band shape or fine structure need to be illustrated, may qualify for this exception. When presentation of spectra is essential, only the pertinent sections should be included, either in the text or as supplementary information (Supplementary Material file).
Crystallographic data. Only data and graphics vital to the discussion should be included in the manuscript. If the data are used solely for confirming compound identity or stereochemistry, a statement in the results and discussion or experimental section that the assignment is supported by an X-ray crystallographic structure determination is usually sufficient, though a ball-and-stick drawing or thermal ellipsoid plot can be included if desired. Regardless of the level of detail of the discussion of the structure, a Crystallographic Information File (CIF) containing complete details of data collection, crystal and unit-cell parameters, structure solution and refinement, and tables of atomic coordinates and thermal parameters, bond lengths, bond angles, and torsion angles should be furnished as supporting information. Reflection intensity data (structure factor tables) should not be included. If more than one crystal structure is being reported, the CIF for each structure should be furnished as a separate file. CIFs should be saved in text-only (plain ASCII) format and should be assigned file names that identify the structure number used in the manuscript and that include a ".cif" (not ".txt") extension. Before being submitted, CIFs should be checked and corrected using the free checkCIF data-validation utility at
http://journals.iucr.org/services/cif/checkcif.html.
All structures for which an X-ray determination is included must also be submitted to the Cambridge Crystallographic Data Base. The unique number assigned to the compound in the data base must be quoted in the experimental description of the compound.
For combinatorial libraries of more than 20 compounds characterization data must be provided for at least 20 diverse members.
Other material including, for example, modeling programs, instrument and circuit diagrams, graphical kinetic data, and non-essential spectroscopic (e.g., IR, NMR, MS and UV/visible) data or other instrumental (e.g., electrochemical or chromatographic) details should be submitted in a separate Supplementary Material File (see Section 3).
2.2.15 Acknowledgements (optional)
2.2.16 References (and Notes)
A separate number must be assigned to each reference cited (e.g., 1-5); groups of references may not be nested under one number (e.g., 1a-e). All references should be listed in the "References" section at the end of the paper in numerical order with no text between the reference number and the first author's surname (e.g., "3. Jones, A..." not "3. See also Jones, A..."). There must be
one blank line between each reference listed. The number of references cited must be limited to 50 for research papers. Papers that exceed this limit will be returned to the authors. Long reference lists should be avoided by selecting references carefully and citing reviews, or selecting recent papers which contain many pertinent older citations, whenever possible. References may appear in the reference list followed by "and references cited therein" if appropriate. There is no limit to the number of references that may be cited in Reviews and Accounts. Reference numbers in the text should be written as
superscript numerals (without brackets), except when the number is integral to the sense; then it is not superscripted (e.g., "see ref. 3"). When placed following a punctuation point (full stop, comma, etc.) they should follow the point directly without a space. A sequence of numbers should be separated by commas, without spaces (e.g., 5,7,8). A consecutive sequence of three or more should be telescoped (e.g., 2-4).
Note: No hyperlinks between superscripts in the text and actual references in the
Reference Section may be used. Brief notes may be used to cite manuscripts in preparation, unpublished observations and personal communications, but longer notes should appear in the Supplementary Material section. Experimental procedures or data must not be included in the reference list.
The Chem. Abstr. reference should be cited for less accessible literature (e.g., patents). Digital Object Identifier (DOI) numbers should be given for papers cited that have been published online, but have yet to receive page numbers. Authors are responsible for obtaining written approval for all personal communications and sending a copy of the manuscript to those cited as authors of personal communications.
Do not cite issue numbers, unless the journal is paginated afresh with each issue. Use standard abbreviations of journal titles.
References cited should follow the style below:
PAPERS
1. Bakke, J. M.; Gautun, H. S. H.; Sletvold, I.
Arkivoc 2001, (
x), 26.
2. Herz, W.; Iyer, V. S.; Gopal Nair, M.
J. Org. Chem. 1975,
40, 3519.
3. Wanner, M. J.; Koomen, G.-J.
J. Chem. Soc., Perkin Trans. 1 2001, 1908.
Although not essential, authors are encouraged to give both the first and last pages of papers (as
shown below) but if this is done it must be done for all the references in the manuscript.
Wanner, M. J.; Koomen, G.-J.
J. Chem. Soc., Perkin Trans. 1 2001, 1908-1915.
BOOKS
1. Henderson, B.; McIndoe, J. S. In
Comprehensive Coordination Chemistry; McCleverty, J. A.;
Meyer, T. J. Eds.; Elsevier: Oxford, 2003; Vol. 1, p 387.
2. Reichardt, C.
Solvents and Solvent Effects in Organic Chemistry; VCH: Weinheim, 1990; Vol.
4, pp 298-311.
3. Furniss, B. S.; Hannaford, A. J.; Smith, P. W. G.; Tatchell, A. R.
Vogel's Textbook of Practical
Organic Chemistry, 5th Edn.; Wiley: New York, 1989; pp 64-67.
DISSERTATIONS
1. Lund, K. P. Ph.D. Thesis, Univ. California, San Diego, 1992.
PATENTS
1. Wright, J. B. U.S. Patent 3 115 496, 1963;
Chem. Abstr. 1964,
60, P 5512b.
2. Baylis, A. B.; Hillman, M. E. D. Ger. Offen. 2 155 113, 1972;
Chem. Abstr. 1972,
77, 34174.
3. Buntain, I. G.; Hatton, L. R. Eur. Pat. Appl. 295 117, 1988;
Chem. Abstr. 1990,
112, P 35845n.
4. Alia, J. M.; Backendorf, C. M. P. PCT Int. Appl. 075 903, 2003;
Chem. Abstr. 2003,
139, 240348.
CONFERENCE PROCEEDINGS
1. Scott, L. T.; Cheng, P.-C.; Bratcher, M. S. Eighth International Symposium on Novel Aromatic
Compounds, Victoria, British Columbia, Canada, July 19-24, 1992: Abstract No. 64.
Note the blank line between references. This is not necessary if authors provide DOI numbers. If this is done, please use this format:
http://dx.doi.org/10.1002/amie.20100296.
Otherwise, the numbers will be supplied by the journal.
2.2.17 Authors' Biographies (only required for reviews and accounts)
Each author should provide a photograph (portrait style, size 2 x 1.5 inches) and a short professional biography at the end of the manuscript.
2.4.1 Schemes, equations, and structure blocks
Scheme: depicts a series of chemical transformations containing chemical structures.
Equation: illustrates a simpler transformation with a single reaction arrow.
Structure Block: a collection of one or more numbered structures.
Wherever possible, all graphics appear in the text immediately following the paragraph in which they are first mentioned.
For preparation of Schemes, Equations, and Structure Blocks, CS ChemDraw is the recommended drawing program.
Do not insert the Schemes, Equations, and Structure Blocks as picture images.
With ChemDraw the following settings should be used:
Text font: Arial, 10 pt
Fixed length: 14.4 pt (0.508 cm, 0.2 in)
Bold width: 2.0 pt (0.071 cm, 0.0278 in)
Line width: 0.6 pt (0.021 cm, 0.0084 in)
Bond spacing: 18% of width
Hash spacing: 2.5 pt (0.088 cm, 0.0347 in)
Owing to the incomplete implementation of the MATH tag in some of the most popular browsers, efficient handling of mathematical equations is not possible. Therefore, authors should create their mathematical equations in a layout program of their choice and then save the equation as an image in jpg format.
Within Schemes, Equations, or Structure Blocks, individual structures may be numbered with boldface Arabic numerals and lower case letters, e.g.,
32a, 32b.
Schemes or chemical equations must be cited in the text using Arabic numerals, e.g., Scheme 1, Equation 1, etc.
Captions should be presented separately from the Scheme and Equation, not incorporated into the block. Note that captions are normally not sentences and need not be followed by a full stop.
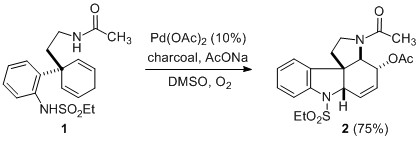
Scheme 1. Pd(II)-catalyzed aza-Wacker cascade.
log k = (-2.62 ± 0.40)σ - (1.69 ± 0.17) (1)
When inserting Schemes, Equations, and Structure Blocks;
do not use the float over text option.
2.4.2 Figures
Diagrams, graphs, spectra, photographs, and any other illustrations should all be considered Figures.
Figures should be labeled sequentially with Arabic numerals as they appear, i.e.,
Figure 1, etc. together with a caption appearing separately below each Figure.
Figures should be included within the body of the manuscript close to the relevant text, but at the end of a paragraph.
Color art 300 dpi
Example:
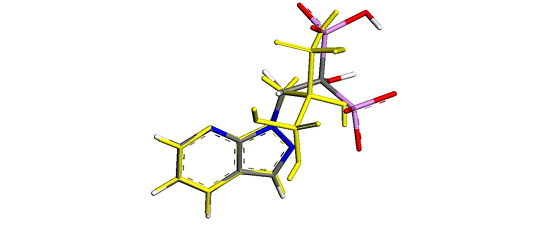 Figure 1.
Figure 1. Lowest energy conformations obtained for BP6 superimposed with the structure obtained experimentally by X-ray crystallography (yellow, C – grey, N – blue, O – red, P – violet, H – white.
4. Submission of Manuscript
4.1 Submission procedure
Manuscripts should be submitted via the Arkivoc online submission site
www.arkat-usa.org/arkivoc-journal/online-submission/. In order to submit, the corresponding author should login or register if using it for the first time. After login, click ‘Submit Manuscript’, read the disclaimer and proceed to Step 1. On the Step 1 form please enter your contact information and add co-authors' names as they appear in the paper masthead. You may add more than one co-author by using the 'Add co-author' button. Please try to ensure that names are not repeated. After completing the first step click on ‘Proceed to step 2’. On the next form you will need to complete the Manuscript Title, select the Manuscript Type and the Issue to which the manuscript belongs. Please type in all the information. Copying and pasting may create invalid characters. To add Greek letters and format text to the title use the appropriate buttons. You must suggest the names of at least 3 referees for review. Step 3 will allow you to upload Manuscript, Graphical Abstract and Supporting Information files. We only accept Microsoft Word (doc or .docx) file format. At Step 4 review your previously inputted information and submit your manuscript.
i. Manuscript - Please use the template provided.
ii. Supplementary Materials Reserved for 1H and 13C NMR spectra, other (bio)chemical spectra, extended tables, etc. See Section 3.
iii. Authors are requested to provide suggestions of names of four referees, and provide the institution, and e-mail address, of each referee.
4.2 Refereeing procedure and editing of the manuscript
All manuscripts approved by the Primary Review Committee will be reviewed by designated Arkivoc referees.
The Scientific Editor will receive reports from the referees. Upon receipt of referees' reports the Scientific Editor will make a decision to accept, reject or refer the manuscript back to the authors.
Proofs of the paper will be sent to the corresponding author for approval prior to publication on the website. Only minor changes can be made at the proof stage.
If in the opinion of the Publishing Editor authors request excessive proof corrections arising from the authors' own errors in the original manuscript then the authors will be required to pay the full cost of the corrections, payable before publication, or the manuscript must be withdrawn and resubmitted.
4.3 Revision of manuscript
If a manuscript needs to be revised, the author should address ALL concerns and suggestions made by referees and/or the Scientific or Publishing editors.
The revised manuscript must be submitted to the Scientific Editor within 30 days. Failing this, the manuscript will be considered to be withdrawn. Resubmission of an unsatisfactorily revised manuscript may result in rejection. If another revision is needed, the time limit is two weeks.
4.4 Copyright agreement
Authors sign an Author Agreement by which they retain copyright but license non-exclusive rights in their article to the Publisher. Creative Commons License Deed; Attribution-NonCommercial 4.0 International (CC BY 4.0)
https://creativecommons.org/licenses/by/4.0/
5. Model Experimental Section
Experimental Section
General.1H NMR and
13C NMR were recorded on a Bruker Avance 300 (
1H: 300 MHz,
13C: 75.5 MHz), Bruker AC-250 FT (
1H: 250 MHz,
13C: 62.9 MHz), using solvent peak as internal reference or apparatus SR. The chemical shifts (δ) and coupling constants (J) are expressed in ppm and hertz respectively. "a" means "apparent" for close coupling constant and the number of equivalent group are indicated after the sign "x". Carbon attribution C, CH, CH
2 and CH
3 were determined by
13C, DEPT 135 and HMQC experiments. InfraRed (IR) spectra were recorded on a Perkin-Elmer Paragon 1000 FT-IR spectrophotometer. Melting points were determined by using a Büchi-Tottoli apparatus. Merck silica gel (0.043-0.063 mm) was used for flash chromatography. CH
2Cl
2 (DCM) and acetonitrile were distilled from CaH
2. All reactions were carried out under nitrogen. Other reagents and starting materials were directly used as obtained commercially.
N-[2-(1-Phenylcyclohexa-2,5-dienyl)ethyl]methanesulfonamide (3c). To a solution of 2-(1-Phenylcyclohexa-2,5-dienyl)-ethylamine
20,21 (536 mg, 2.69 mmol) in DCM (26 mL) was added triethylamine (760 μL, 5.42 mmol) and methanesulfonyl chloride (210 μL, 2.71 mmol) at 0 °C. After stirring overnight at room temperature, the organic layer was washed with water then brine, dried over anhydrous sodium sulfate and filtered. Evaporation of the solvents gave a colorless solid which was purified through silica gel chromatography (Petroleum ether/EtOAc 80:20) to give
N-[2-(1-phenylcyclohexa-2,5-dienyl)ethyl]methanesulfonamide
3c as a colorless solid (737 mg, 98%). mp 94-96 °C. IR (solid, KBr, νmax, cm
-1): 3288, 1314, 1153, 1060, 728, 703, 595, 525, 514.
1H NMR (300 MHz, CDCl
3): δ
H 2.10-2.15 (2H, m, CH
2CH
2N), 2.68-2.74 (2H, m, bis allylic CH
2), 2.94 (3H, s, NSO
2CH
3), 3.15-3.22 (2H, m, CH
2N), 4.83 (1H, broad s, NH), 5.61-5.66 (2H, m, CH x 2 olefinic), 5.92 (2H, dt
a,
3JHH 10.5,
4JHH 3.4 and 3.4 Hz, CH x 2 olefinic), 7.17-7.23 (1H, m, CH aromatic), 7.29-7.36 (4H, m, 4CH aromatic).
13C NMR (CDCl
3, 75.5 MHz): δ
C 26.0 (CH
2 bis-allylic), 39.8 (CH
2CH
2N), 40.1 (NSO
2CH
3), 40.8 (CH
2N), 43.0 (C aliphatic), 124.7 (CH x 2, olefinic), 126.4 (CH aromatic), 126.4, 128.5 (2CH x 2 aromatic), 131.7 (CH x 2 olefinic), 147.1 (C aromatic). Anal. calcd for C
15H
19NO
2S (277.11): C, 64.95; H, 6.90; N, 5.05; S, 11.56. Found: C, 65.17; H, 7.18; N, 4.88; S, 11.79.
[These instructions approved by the Arkivoc Steering Committee 19th November 2023]